Abstract
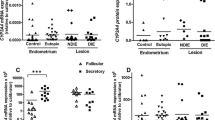
We have shown previously that high expression levels of microsomal epoxide hydrolase (mEH) correlate with a poor prognosis of breast cancer patients receiving tamoxifen, suggesting that enhanced mEH expression could lead to antiestrogen resistance (Fritz et al. in J Clin Oncol 19:3–9, 2001). Thus, the purpose of this study was to gain insights into the role of mEH in hormone-responsive tissues. We analyzed biopsy samples of the endometrium by immunohistochemical staining, pointing to a regulation of mEH during the menstrual cycle: during the first half mEH expression was low, increased during the second half and reached highest levels during pregnancy. Additionally, the progesterone receptor (PR) positive human endometrial cell lines IKPRAB-36 (estrogene receptor α [ERα] negative) and ECC1-PRAB72 (ERα positive) were chosen to further investigate the hormonal regulation of mEH expression. Western Blot and quantitative RT-PCR analysis revealed an increase of mEH expression after treatment with medroxy-progesterone 17-acetate (MPA) in the ERα containing ECC1-PRAB72 cells. In contrast our results suggest that MPA had no influence on the mEH protein level in the ERα- IKPRAB-36 cells. In conclusion, mEH expression is regulated by progesterone in the presence of both PRs and ERα.





Similar content being viewed by others
References
Ballare C, Uhrig M, Bechtold T, Sancho E, Di DM, Migliaccio A et al (2003) Two domains of the progesterone receptor interact with the estrogen receptor and are required for progesterone activation of the c-Src/Erk pathway in mammalian cells. Mol Cell Biol 23:1994–2008
Beck T, Weikel W, Brumm C, Wilkens C, Pollow K, Knapstein PG (1994) Immunohistochemical detection of hormone receptors in breast carcinomas (ER-ICA, PgR-ICA): prognostic usefulness and comparison with the biochemical radioactive-ligand-binding assay (DCC). Gynecol Oncol 53:220–227
Berthois Y, Katzenellenbogen JA, Katzenellenbogen BS (1986) Phenol red in tissue culture media is a weak estrogen: implications concerning the study of estrogen-responsive cells in culture. Proc Natl Acad Sci U S A 83:2496–2500
Butts CL, Shukair SA, Duncan KM, Bowers E, Horn C, Belyavskaya E et al (2007) Progesterone inhibits mature rat dendritic cells in a receptor-mediated fashion. Int Immunol 19:287–296
Coezy E, Borgna JL, Rochefort H (1982) Tamoxifen and metabolites in MCF7 cells: correlation between binding to estrogen receptor and inhibition of cell growth. Cancer Res 42:317–323
Coller JK, Fritz P, Zanger UM, Siegle I, Eichelbaum M, Kroemer HK et al (2001) Distribution of microsomal epoxide hydrolase in humans: an immunohistochemical study in normal tissues, and benign and malignant tumours. Histochem J 33:329–336
Cresteil T, Beaune P, Kremers P, Celier C, Guengerich FP, Leroux JP (1985) Immunoquantification of epoxide hydrolase and cytochrome P-450 isozymes in fetal and adult human liver microsomes. Eur J Biochem 151:345–350
de Medina P, Payre B, Boubekeur N, Bertrand-Michel J, Terce F, Silvente-Poirot S et al (2009a) Ligands of the antiestrogen-binding site induce active cell death and autophagy in human breast cancer cells through the modulation of cholesterol metabolism. Cell Death Differ 16:1372–1384
de Medina P, Silvente-Poirot S, Poirot M (2009b) Tamoxifen and AEBS ligands induced apoptosis and autophagy in breast cancer cells through the stimulation of sterol accumulation. Autophagy 5:1066–1067
de Waziers I, Cugnenc PH, Yang CS, Leroux JP, Beaune PH (1990) Cytochrome P 450 isoenzymes, epoxide hydrolase and glutathione transferases in rat and human hepatic and extrahepatic tissues. J Pharmacol Exp Ther 253:387–394
Decker M, Arand M, Cronin A (2009) Mammalian epoxide hydrolases in xenobiotic metabolism and signalling. Arch Toxicol 83:297–318
Denlinger CL, Vesell ES (1989) Hormonal regulation of the developmental pattern of epoxide hydrolases. Studies in rat liver. Biochem Pharmacol 38:603–610
Fabian C, Tilzer L, Sternson L (1981) Comparative binding affinities of tamoxifen, 4-hydroxytamoxifen, and desmethyltamoxifen for estrogen receptors isolated from human breast carcinoma: correlation with blood levels in patients with metastatic breast cancer. Biopharm Drug Dispos 2:381–390
Fargin A, Faye JC, le MM, Bayard F, Potier M, Beauregard G (1988) Solubilization of a tamoxifen-binding protein. Assessment of its molecular mass. Biochem J 256:229–236
Farin FM, Omiecinski CJ (1993) Regiospecific expression of cytochrome P-450 s and microsomal epoxide hydrolase in human brain tissue. J Toxicol Environ Health 40:317–335
Faye JC, Jozan S, Redeuilh G, Baulieu EE, Bayard F (1983) Physicochemical and genetic evidence for specific antiestrogen binding sites. Proc Natl Acad Sci U S A 80:3158–3162
Fritz P, Behrle E, Zanger UM, Murdter T, Schwarzmann P, Kroemer HK (1996) Immunohistochemical assessment of human microsomal epoxide hydrolase in primary and secondary liver neoplasm: a quantitative approach. Xenobiotica 26:107–116
Fritz P, Murdter TE, Eichelbaum M, Siegle I, Weissert M, Zanger UM (2001) Microsomal epoxide hydrolase expression as a predictor of tamoxifen response in primary breast cancer: a retrospective exploratory study with long-term follow-up. J Clin Oncol 19:3–9
Guengerich FP (1994) Epoxide hydrolase: properties and metabolic roles. Rev Biochem Toxicol 4:5–30
Guenthner TM (1990) Epoxide hydrolase. In: Mulder GJ (ed) Conjugation reactions in drug metabolism. Taylor & Francis, London, pp 365–404
Hammock BD, Storms DH, Grant DF (1997) Epoxide hydrolases. In: Guengerich FP (ed) Comprehensive toxicology, vol 3. Pergamon, Oxford, pp 283–305
Hanifi-Moghaddam P, Sijmons B, Ott MC, van Ijcken WF, Nowzari D, Kuhne EC et al (2006) The hormone replacement therapy drug tibolone acts very similar to medroxyprogesterone acetate in an estrogen-and progesterone-responsive endometrial cancer cell line. J Mol Endocrinol 37:405–413
Inoue N, Yamada K, Imai K, Aimoto T (1993) Sex hormone-related control of hepatic epoxide hydrolase activities in mice. Biol Pharm Bull 16:1004–1007
Jackson MR, Craft JA, Burchell B (1987) Nucleotide and deduced amino acid sequence of human liver microsomal epoxide hydrolase. Nucleic Acids Res 15:7188
Jordan VC (1984) Biochemical pharmacology of antiestrogen action. Pharmacol Rev 36:245–276
Katzenellenbogen BS (2000) Mechanisms of action and cross-talk between estrogen receptor and progesterone receptor pathways. J Soc Gynecol Investig 7:S33–S37
Kedjouar B, Daunes S, Vilner BJ, Bowen WD, Klaebe A, Faye JC et al (1999) Structural similitudes between cytotoxic antiestrogen-binding site (AEBS) ligands and cytotoxic sigma receptor ligands. Evidence for a relationship between cytotoxicity and affinity for AEBS or sigma-2 receptor but not for sigma-1 receptor. Biochem Pharmacol 58:1927–1939
Kedjouar B, de MP, Oulad-Abdelghani M, Payre B, Silvente-Poirot S, Favre G et al (2004) Molecular characterization of the microsomal tamoxifen binding site. J Biol Chem 279:34048–34061
Kim SK, Woodcroft KJ, Kim SG, Novak RF (2003) Insulin and glucagon signaling in regulation of microsomal epoxide hydrolase expression in primary cultured rat hepatocytes. Drug Metab Dispos 31:1260–1268
Kon OL (1983) An antiestrogen-binding protein in human tissues. J Biol Chem 258:3173–3177
Labit-Le Bouteiller C, Jamme MF, David M, Silve S, Lanau C, Dhers C et al (1998) Antiproliferative effects of SR31747A in animal cell lines are mediated by inhibition of cholesterol biosynthesis at the sterol isomerase step. Eur J Biochem 256:342–349
Levin W, Michaud DP, Thomas PE, Jerina DM (1983) Distinct rat hepatic microsomal epoxide hydrolases catalyze the hydration of cholesterol 5, 6 alpha-oxide and certain xenobiotic alkene and arene oxides. Arch Biochem Biophys 220:485–494
Mesange F, Sebbar M, Kedjouar B, Capdevielle J, Guillemot JC, Ferrara P et al (1998) Microsomal epoxide hydrolase of rat liver is a subunit of theanti-oestrogen-binding site. Biochem J 334(Pt 1):107–112
Mesange F, Sebbar M, Capdevielle J, Guillemot JC, Ferrara P, Bayard F et al (2002) Identification of two tamoxifen target proteins by photolabeling with 4-(2-morpholinoethoxy)benzophenone. Bioconjug Chem 13:766–772
Miller MA, Katzenellenbogen BS (1983) Characterization and quantitation of antiestrogen binding sites in estrogen receptor- positive and negative human breast cancer cell lines. Cancer Res 43:3094–3100
Mitsushita J, Toki T, Kato K, Fujii S, Konishi I (2000) Endometrial carcinoma remaining after term pregnancy following conservative treatment with medroxyprogesterone acetate. Gynecol Oncol 79:129–132
Moebius FF, Reiter RJ, Bermoser K, Glossmann H, Cho SY, Paik YK (1998) Pharmacological analysis of sterol delta8-delta7 isomerase proteins with [3H]ifenprodil. Mol Pharmacol 54:591–598
Montgomery RV (2002) Hormone replacement therapy: optimising the dose and route of administration. Drugs Aging 19:807–818
Murray GI, Taylor VE, McKay JA, Weaver RJ, Ewen SW, Melvin WT et al (1995) Expression of xenobiotic metabolizing enzymes in tumours of the urinary bladder. Int J Exp Pathol 76:271–276
Oesch F (1974) Purification and specificity of a human microsomal epoxide hydratase. Biochem J 139:77–88
Oesch F (1983) Drug detoxification: epoxide hydrolase. Prog Clin Biol Res 135:81–105
Omiecinski CJ, Aicher L, Swenson L (1994) Developmental expression of human microsomal epoxide hydrolase. J Pharmacol Exp Ther 269:417–423
Paul R, Silve S, De Nys N, Dupuy PH, Bouteiller CL, Rosenfeld J et al (1998) Both the immunosuppressant SR31747 and the antiestrogen tamoxifen bind to an emopamil-insensitive site of mammalian Delta8-Delta7 sterol isomerase. J Pharmacol Exp Ther 285:1296–1302
Payre B, de Medina P, Boubekeur N, Mhamdi L, Bertrand-Michel J, Terce F et al (2008) Microsomal antiestrogen-binding site ligands induce growth control and differentiation of human breast cancer cells through the modulation of cholesterol metabolism. Mol Cancer Ther 7:3707–3718
Remmele W, Stegner HE (1987) Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 8:138–140
Seidegard J, DePierre JW (1983) Microsomal epoxide hydrolase. Properties, regulation and function. Biochim Biophys Acta 695:251–270
Skoda RC, Demierre A, McBride OW, Gonzalez FJ, Meyer UA (1988) Human microsomal xenobiotic epoxide hydrolase. Complementary DNA sequence, complementary DNA-directed expression in COS-1 cells, and chromosomal localization. J Biol Chem 263:1549–1554
Sudo K, Monsma FJ Jr, Katzenellenbogen BS (1983) Antiestrogen-binding sites distinct from the estrogen receptor: subecellular localization, ligand specificity, and distribution in tissues of the rat. Endocrinology 112:425–434
Sutherland RL, Murphy LC, San FM, Green MD, Whybourne AM, Krozowski ZS (1980) High-affinity anti-oestrogen binding site distinct from the oestrogen receptor. Nature 288:273–275
Thomas H, Schladt L, Knehr M, Oesch F (1989) Effect of diabetes and starvation on the activity of rat liver epoxide hydrolases, glutathione S-transferases and peroxisomal beta-oxidation. Biochem Pharmacol 38:4291–4297
Vogel-Bindel U, Bentley P, Oesch F (1982) Endogenous role of microsomal epoxide hydrolase. Ontogenesis, induction inhibition, tissue distribution, immunological behaviour and purification of microsomal epoxide hydrolase with 16 alpha, 17 alpha-epoxyandrostene-3-one as substrate. Eur J Biochem 126:425–431
von Dippe P, Amoui M, Stellwagen RH, Levy D (1996) The functional expression of sodium-dependent bile acid transport in Madin-Darby canine kidney cells transfected with the cDNA for microsomal epoxide hydrolase. J Biol Chem 271:18176–18180
Watts CK, Sutherland RL (1984) High affinity specific antiestrogen binding sites are concentrated in rough microsomal membranes of rat liver. Biochem Biophys Res Commun 120:109–115
Watts CK, Murphy LC, Sutherland RL (1984) Microsomal binding sites for nonsteroidal anti-estrogens in MCF 7 human mammary carcinoma cells. Demonstration of high affinity and narrow specificity for basic ether derivatives of triphenylethylene. J Biol Chem 259:4223–4229
Acknowlegments
We thank Stefanie Laukemann and Susanne Gutzeit for excellent technical assistance and Christian Joffroy for the GUS primers and the RT-PCR protocol.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Popp, S.L., Abele, I.S., Buck, M.B. et al. Microsomal epoxide hydrolase expression in the endometrial uterine corpus is regulated by progesterone during the menstrual cycle. J Mol Hist 41, 111–119 (2010). https://doi.org/10.1007/s10735-010-9266-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-010-9266-6