Abstract
Introduction
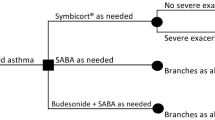
In budesonide/formoterol (Symbicort® Turbuhaler®, AstraZeneca, Lund, Sweden) maintenance and reliever therapy (SMART), patients with asthma take a daily maintenance dose of budesonide/formoterol, with the option of taking additional doses for symptom relief instead of a short-acting β2-agonist (SABA). This study assesses the cost-effectiveness of SMART compared with usual care in patients with mild-to-moderate persistent asthma treated by general practitioners in the Netherlands from a societal perspective.
Methods
The study was linked to a randomized, active-controlled, open-label, multicenter, 12-month clinical trial, with a prospective collection of resource use. One hundred and two patients ≥18 years with mild-to-moderate persistent asthma and daily inhaled corticosteroids (ICS) prior to the trial were included. SMART was given as two inhalations of budesonide/formoterol (100/6 μg) once daily, plus additional doses as needed. The control group was treated according to guidelines, which prescribe medium daily doses of ICS plus an SABA if needed. A long-acting β2-agonist (LABA) is added if necessary. Effectiveness was measured as the proportion of asthma-control days, Asthma Control Questionnaire (ACQ) scores, the net proportion of patients with relevant ACQ improvement, and the proportion of well-controlled patients. Costs included asthma medication, physician contacts, and absence from work.
Results
Mean total costs for SMART were €134.81 lower (95% CI: −€439.48; €44.85). Production losses were €94.10 (95% CI: −€300.60; €0.29) lower for SMART (€10.77 vs. €104.87). No significant differences in health outcomes were seen, with 3.81 fewer asthma-control days per patient-year for SMART (95% CI: −36.8; 30.8), a 0.049 better ACQ score (95% CI: −0.21; 0.29), a 5.8% larger net proportion of improved patients (95% CI: t15.6%; 27.3%), and a 2.1% (95% CI: −25.5; 20.8%) smaller increase in the proportion of well-controlled patients.
Conclusions
Treating primary care patients with mild-to-moderate persistent asthma with SMART instead of ICS plus bronchodilators does not affect health outcomes and does not increase costs; therefore, is likely to be an alternative for guideline-directed treatment, from a health and economic perspective.
Similar content being viewed by others
References
Global Initiative for Asthma web site. GINA Report, Global Strategy for Asthma Management and Prevention, Global Initiative for Asthma 2008. Available at: www.ginasthma.com. Accessed August 2009.
Kips JC, O’Connor BJ, Inman MD, Svensson K, Pauwels RA, O’Byrne PM. A long-term study of the antiinflammatory effect of low-dose budesonide plus formoterol versus high-dose budesonide in asthma. Am J Respir Crit Care Med. 2000;161:996–1001.
Lalloo UG, Malolepszy J, Kozma D, et al. Budesonide and formoterol in a single inhaler improves asthma control compared with increasing the dose of corticosteroid in adults with mild-to-moderate asthma. Chest. 2003;123:1480–1487.
Stoloff SW, Stempel DA, Meyer J, Stanford RH, Carranza Rosenzweig JR. Improved refill persistence with fluticasone propionate and salmeterol in a single inhaler compared with other controller therapies. J Allergy Clin Immunol. 2004;113:245–251.
O’Byrne PM, Bisgaard H, Godard PP, et al. Budesonide/formoterol combination therapy as both maintenance and reliever medication in asthma. Am J Respir Crit Care Med. 2005;171:129–136.
Rabe KF, Atienza T, Magyar P, Larsson P, Jorup C, Lalloo UG. Effect of budesonide in combination with formoterol for reliever therapy in asthma exacerbations: a randomised controlled, doubleblind study. Lancet. 2006;368:744–753.
Rabe KF, Pizzichini E, Stallberg B, et al. Budesonide/formoterol in a single inhaler for maintenance and relief in mild-to-moderate asthma: a randomized, double-blind trial. Chest. 2006;129:246–256.
Scicchitano R, Aalbers R, Ukena D, et al. Efficacy and safety of budesonide/formoterol single inhaler therapy versus a higher dose of budesonide in moderate to severe asthma. Curr Med Res Opin. 2004;20:1403–1418.
Vogelmeier C, D’Urzo A, Pauwels R, et al. Budesonide/formoterol maintenance and reliever therapy: an effective asthma treatment option? Eur Respir J. 2005;26:819–828.
Johansson G, Andreasson EB, Larsson PE, Vogelmeier CF. Cost effectiveness of budesonide/formoterol for maintenance and reliever therapy versus salmeterol/fluticasone plus salbutamol in the treatment of asthma. Pharmacoeconomics. 2006;24:695–708.
Lundborg M, Wille S, Bjermer L, et al. Maintenance plus reliever budesonide/formoterol compared with a higher maintenance dose of budesonide/formoterol plus formoterol as reliever in asthma: an efficacy and cost-effectiveness study. Curr Med Res Opin. 2006;22:809–821.
Price D, Wiren A, Kuna P. Cost-effectiveness of budesonide/formoterol for maintenance and reliever asthma therapy. Allergy. 2007;62:1189–1198.
Miller E, Sears MR, McIvor A, Liovas A. Canadian economic evaluation of budesonide-formoterol as maintenance and reliever treatment in patients with moderate to severe asthma. Can Respir J. 2007;14:269–275.
Ställberg B, Ekstrom T, Neij F, et al. A real-life costeffectiveness evaluation of budesonide/formoterol maintenance and reliever therapy in asthma. Respir Med. 2008;102:1360–1370.
Geijer RMM, Chavannes NH, Muris JWM, et al. NHG Standaard Astma bij volwassenen. Huisarts en Wetenschap. 2007;11:537–551.
Juniper EF, O’Byrne PM, Roberts JN. Measuring asthma control in group studies: do we need airway calibre and rescue beta2-agonist use? Respir Med. 2001;95:319–323.
Juniper EF, O’Byrne PM, Guyatt GH, Ferrie PJ, King DR. Development and validation of a questionnaire to measure asthma control. Eur Respir J. 1999;14:902–907.
Campbell JL, Kiebert GM, Partridge MR. Development of the satisfaction with inhaled asthma treatment questionnaire. Eur Respir J. 2003;22:127–134.
Guyatt GH, Juniper EF, Walter SD, Griffith LE, Goldstein RS. Interpreting treatment effects in randomised trials. BMJ. 1998;316:690–693.
Juniper EF, Bousquet J, Abetz L, Bateman ED, GOAL Committee. Identifying “well-controlled” and “not well-controlled” asthma using the Asthma Control Questionnaire. Respir Med. 2006;100:616–621.
Oostenbrink JB, Bouwmans CAM, Koopmanschap MA, Rutten FFH. Guide to cost research - Methods and standard costs prices for economic evaluatons in healthcare [in Dutch]. Amstelveen: College voor zorgverzekeringen (Health Care Insurance Board); 2004.
Health Care Insurance Board. Available at: www.medicijnkosten.nl (Medication costs). Accessed September 10, 2007.
Briggs AH, Wonderling DE, Mooney CZ. Pulling cost-effectiveness analysis up by its bootstraps: a non-parametric approach to confidence interval estimation. Health Econ. 1997;6:327–340.
Campbell MK, Torgerson DJ. Bootstrapping: estimating confidence intervals for cost-effectiveness ratios. QJM. 1999;92:177–182.
DiCiccio TJ, Efron B. Bootstrap confidence intervals. Stat Sci. 1996;11:189–228.
Briggs A, Fenn P. Confidence intervals or surfaces? Uncertainty on the cost-effectiveness plane. Health Econ. 1998;7:723–740.
Briggs AH, O’Brien BJ. The death of costminimization analysis? Health Econ. 2001;10:179–184.
Al MJ, van Hout BA, Michel BC, Rutten FF. Sample size calculation in economic evaluations. Health Econ. 1998;7:327–335.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goossens, L.M.A., Riemersma, R.A., Postma, D.S. et al. An economic evaluation of budesonide/formoterol for maintenance and reliever treatment in asthma in general practice. Adv Therapy 26, 872–885 (2009). https://doi.org/10.1007/s12325-009-0063-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-009-0063-0