2018-01-16
Characterization of Endothelial Cells Associated with Hematopoietic Niche Formation in Humans Identifies IL-33 As an Anabolic Factor
Publication
Publication
Cell Reports , Volume 22 - Issue 3 p. 666- 678
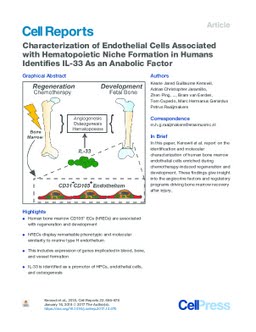
Bone marrow formation requires an orchestrated interplay between osteogenesis, angiogenesis, and hematopoiesis that is thought to be mediated by endothelial cells. The nature of the endothelial cells and the molecular mechanisms underlying these events remain unclear in humans. Here, we identify a subset of endoglin-expressing endothelial cells enriched in human bone marrow during fetal ontogeny and upon regeneration after chemotherapeutic injury. Comprehensive transcriptional characterization by massive parallel RNA sequencing of these cells reveals a phenotypic and molecular similarity to murine type H endothelium and activation of angiocrine factors implicated in hematopoiesis, osteogenesis, and angiogenesis. Interleukin-33 (IL-33) was significantly overexpressed in these endothelial cells and promoted the expansion of distinct subsets of hematopoietic precursor cells, endothelial cells, as well as osteogenic differentiation. The identification and molecular characterization of these human regeneration-associated endothelial cells is thus anticipated to instruct the discovery of angiocrine factors driving bone marrow formation and recovery after injury. In this paper, Kenswil et al. report on the identification and molecular characterization of human bone marrow endothelial cells enriched during chemotherapy-induced regeneration and development. These findings give insight into the angiocrine factors and regulatory programs driving bone marrow recovery after injury.
| Additional Metadata | |
|---|---|
| , , , , , , , | |
| doi.org/10.1016/j.celrep.2017.12.070, hdl.handle.net/1765/104542 | |
| Cell Reports | |
|
Kenswil, K., Jaramillo, A.C. (Adrian Christopher), Ping, Z., Chen, S., Hoogenboezem, R., Mylona, M.-A., … Raaijmakers, M. (2018). Characterization of Endothelial Cells Associated with Hematopoietic Niche Formation in Humans Identifies IL-33 As an Anabolic Factor. Cell Reports, 22(3), 666–678. doi:10.1016/j.celrep.2017.12.070 |
|