2018-05-15
Long-Term Persistence of Exhausted CD8 T Cells in Chronic Infection Is Regulated by MicroRNA-155
Publication
Publication
Cell Reports , Volume 23 - Issue 7 p. 2142- 2156
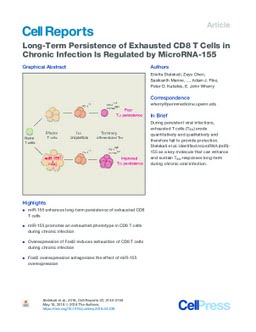
Persistent viral infections and tumors drive development of exhausted T (TEX) cells. In these settings, TEX cells establish an important host-pathogen or host-tumor stalemate. However, TEX cells erode over time, leading to loss of pathogen or cancer containment. We identified microRNA (miR)-155 as a key regulator of sustained TEX cell responses during chronic lymphocytic choriomeningitis virus (LCMV) infection. Genetic deficiency of miR-155 ablated CD8 T cell responses during chronic infection. Conversely, enhanced miR-155 expression promoted expansion and long-term persistence of TEX cells. However, rather than strictly antagonizing exhaustion, miR-155 promoted a terminal TEX cell subset. Transcriptional profiling identified coordinated control of cell signaling and transcription factor pathways, including the key AP-1 family member Fosl2. Overexpression of Fosl2 reversed the miR-155 effects, identifying a link between miR-155 and the AP-1 transcriptional program in regulating TEX cells. Thus, we identify a mechanism of miR-155 regulation of TEX cells and a key role for Fosl2 in T cell exhaustion. During persistent viral infections, exhausted T cells (TEX) erode quantitatively and qualitatively and therefore fail to provide protection. Stelekati et al. identified microRNA (miR)-155 as a key molecule that can enhance and sustain TEX responses long-term during chronic viral infection.
| Additional Metadata | |
|---|---|
| , , , , , , , | |
| doi.org/10.1016/j.celrep.2018.04.038, hdl.handle.net/1765/106542 | |
| Cell Reports | |
| Organisation | Department of Immunology |
|
Stelekati, E. (Erietta), Chen, Z. (Zeyu), Manne, S. (Sasikanth), Kurachi, M. (Makoto), Ali, M.-A. (Mohammed-Alkhatim), Lewy, K. (Keith), … Wherry, E.J. (E. John). (2018). Long-Term Persistence of Exhausted CD8 T Cells in Chronic Infection Is Regulated by MicroRNA-155. Cell Reports, 23(7), 2142–2156. doi:10.1016/j.celrep.2018.04.038 |
|